WISP-1 in Obesity and Metabolic Dysfunction: The Modulatory Role of Exercise Training- A Narrative Review
Keywords:
physical activity, , WISP-1, metabolic health, extracellular matrix, obesity, inflammationAbstract
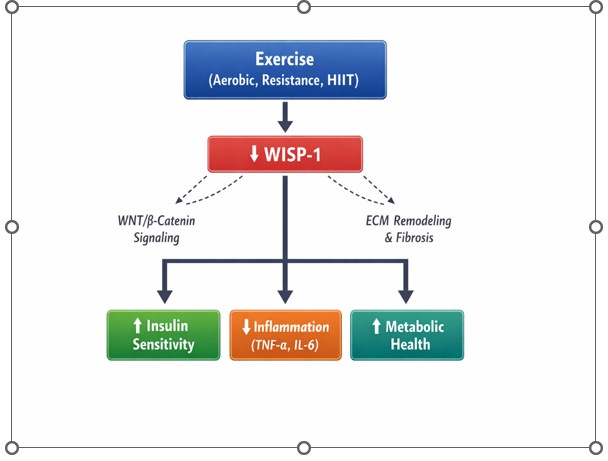
Obesity is a major global health concern strongly associated with metabolic disorders, chronic low-grade inflammation, insulin resistance, and an increased risk of cardiovascular disease. Among the emerging biomarkers implicated in obesity-related metabolic dysfunction, Wnt1-inducible signaling pathway protein-1 (WISP-1), a matricellular protein belonging to the CCN (Cyr61, C: Cyr61(Cysteine-rich angiogenic inducer 61/ C: CTGF (Connective Tissue Growth Factor), N: NOV (Nephroblastoma Overexpressed gene) family, has attracted increasing attention. WISP-1 is involved in tissue remodeling, extracellular matrix regulation, inflammatory signaling, and metabolic processes, and accumulating evidence suggests that elevated circulating levels are associated with visceral adiposity, insulin resistance, and systemic inflammation. Physical activity is widely recognized as a cornerstone intervention for improving metabolic health and reducing obesity-related complications. Exercise training modulates adipose tissue function, inflammatory pathways, insulin signaling, and extracellular matrix remodeling mechanisms that may be linked to alterations in WISP-1 expression. However, the extent to which exercise directly regulates WISP-1 remains incompletely understood, and existing findings are not entirely consistent across populations and intervention types. This narrative review synthesizes current evidence regarding the relationship between physical activity and WISP-1 levels in individuals with obesity. Available studies suggest that aerobic training, resistance exercise, and high-intensity interval training (HIIT) may reduce circulating WISP-1 concentrations, often in parallel with improvements in insulin sensitivity, body composition, and inflammatory markers. Nevertheless, the number of interventional studies remains limited, sample sizes are generally small, and methodological heterogeneity complicates definitive interpretation. Furthermore, much of the mechanistic understanding linking WISP-1 to insulin resistance derives from experimental or associative human data, highlighting the need for cautious interpretation of causality. Variations in exercise modality, intensity, duration, and participant characteristics may influence WISP-1 responses, underscoring the complexity of its regulation. Overall, while emerging evidence supports a potential link between exercise-induced metabolic improvements and modulation of WISP-1, well-designed longitudinal and mechanistic studies are required to clarify its physiological role and determine whether WISP-1 can serve as a reliable biomarker or therapeutic target in obesity related metabolic dysfunction.
Downloads
References
1. Organization WH. Obese and overweight. 2024.
2. Tayebi SM, Ghanbari-Niaki A, Saeidi A, Hackney AC. Exercise training, neuregulin 4 and obesity. Annals of applied sport science. 2017;5(2):1. [PMID: 30899900] [PMCID: PMC6424364] [DOI]
3. Saeidi A, Tayebi SM, Khosravi A, Malekian F, Khodamoradi A, Sellami M, et al. Effects of exercise training on type 2-diabetes: the role of Meteorin-like protein. Health promotion perspectives. 2019;9(2):89. [PMID: 31249794] [PMCID: PMC6588808] [DOI]
4. Saeidi A, Nouri-Habashi A, Razi O, Ataeinosrat A, Rahmani H, Mollabashi SS, et al. Astaxanthin supplemented with high-intensity functional training decreases adipokines levels and Cardiovascular Risk factors in men with obesity. Nutrients. 2023;15(2):286. [PMID: 36678157] [PMCID: PMC9866205] [DOI]
5. Saeidi A, Jabbour G, Ahmadian M, Abbassi-Daloii A, Malekian F, Hackney AC, et al. Independent and combined effects of antioxidant supplementation and circuit resistance training on selected adipokines in postmenopausal women. Frontiers in physiology. 2019;10:484. [PMID: 31105587] [PMCID: PMC6499001] [DOI]
6. Delfan M, Amadeh Juybari R, Gorgani-Firuzjaee S, Høiriis Nielsen J, Delfan N, Laher I, et al. High-intensity interval training improves cardiac function by miR-206 dependent HSP60 induction in diabetic rats. Frontiers in Cardiovascular Medicine. 2022;9:927956. [PMID: 35845054] [PMCID: PMC9277013] [DOI]
7. Ouchi N, Parker JL, Lugus JJ, Walsh K. Adipokines in inflammation and metabolic disease. Nature reviews immunology. 2011;11(2):85-97. [PMID: 21252989] [PMCID: PMC3518031] [DOI]
8. Jun J-I, Lau LF. Taking aim at the extracellular matrix: CCN proteins as emerging therapeutic targets. Nature reviews Drug discovery. 2011;10(12):945-63. [PMID: 22129992] [PMCID: PMC3663145] [DOI]
9. Mirr M, Owecki M. An update to the WISP-1/CCN4 role in obesity, insulin resistance and diabetes. Medicina. 2021;57(2):100. [PMID: 33498604] [PMCID: PMC7911315] [DOI]
10. Pedersen BK, Saltin B. Exercise as medicine–evidence for prescribing exercise as therapy in 26 different chronic diseases. Scandinavian journal of medicine & science in sports. 2015;25:1-72. [PMID: 26606383] [PMCID: PMC12189298] [DOI]
11. Tayebi SM, Saeidi A, Shahghasi R, Golmohammadi M. The eight-week circuit resistance training decreased the serum levels of WISP-1 and WISP-2 in individuals with type 2 diabetes. Annals of Applied Sport Science. 2023;11(4):0-. [PMCID: PMC12694805] [DOI]
12. Chang JS, Kim TH, Kong ID. Exercise intervention lowers aberrant serum WISP-1 levels with insulin resistance in breast cancer survivors: A randomized controlled trial. Scientific reports. 2020;10(1):10898. [PMID: 32616883] [PMCID: PMC7331642] [DOI]
13. Gholami M. Changes of WNT1-inducible signaling pathway protein-1 (WISP-1), ICAM-1 and VCAM-1 in type 2 diabetic women following 12-week high intensity interval training and moderate intensity continuous training. Daneshvar Medicine. 2023;31(3):68-80.
14. Barchetta I, Cimini FA, Capoccia D, De Gioannis R, Porzia A, Mainiero F, et al. WISP1 is a marker of systemic and adipose tissue inflammation in dysmetabolic subjects with or without type 2 diabetes. Journal of the Endocrine Society. 2017;1(6):660-70. [PMID: 29264519] [PMCID: PMC5686652] [DOI]
15. Klimontov VV, Bulumbaeva DM, Fazullina ON, Lykov AP, Bgatova NP, Orlov NB, et al. Circulating Wnt1-inducible signaling pathway protein-1 (WISP-1/CCN4) is a novel biomarker of adiposity in subjects with type 2 diabetes. Journal of Cell Communication and Signaling. 2020;14:101-9. [PMID: 31782053] [PMCID: PMC7176766] [DOI]
16. Habib NS, EL-Hefnawy MH, El-Mesallamy HO. Assessment of circulating Wnt1-inducible signaling pathway protein 1 (WISP1) in obesity and type 2 diabetes mellitus patients. Archives of Pharmaceutical Sciences Ain Shams University. 2018;2(1):47-53. [DOI]
17. Saeidi A, Hackney AC, Tayebi SM, Ahmadian M, Zouhal H. Diabetes, insulin resistance, fetuin-B and exercise training. Annals of applied sport science. 2019;7(2):1-2. [PMID: 31321377] [PMCID: PMC6639044] [DOI]
18. Saeidi A, Seifi-Ski-Shahr F, Soltani M, Daraei A, Shirvani H, Laher I, et al. Resistance training, gremlin 1 and macrophage migration inhibitory factor in obese men: a randomised trial. Archives of Physiology and Biochemistry. 2023;129(3):640-8. [PMID: 33370549] [DOI]
19. Maiese K. WISP1: Clinical insights for a proliferative and restorative member of the CCN family. Current Neurovascular Research. 2014;11(4):378-89. [PMID: 25219658] [PMCID: PMC4205162] [DOI]
20. González D, Campos G, Pütter L, Friebel A, Holland CH, Holländer L, et al. Role of WISP1 in Stellate Cell Migration and Liver Fibrosis. Cells. 2024;13(19):1629. [PMID: 39404393] [PMCID: PMC11475959] [DOI]
21. Christopoulou M-E, Aletras AJ, Papakonstantinou E, Stolz D, Skandalis SS. WISP1 and macrophage migration inhibitory factor in respiratory inflammation: Novel insights and therapeutic potentials for asthma and copd. International Journal of Molecular Sciences. 2024;25(18):10049. [PMID: 39337534] [PMCID: PMC11432718] [DOI]
22. Hörbelt T. The role of the novel adipokines WISP1 and sFRP4 in obesity and type 2 diabetes and their impact on insulin action and energy metabolism in muscle and liver: Dissertation, Düsseldorf, Heinrich-Heine-Universität, 2018; 2018.
23. Grünberg J. WISP-2-A Novel Adipokine Related to Obesity and Insulin resistance2015.
24. Singh K, Oladipupo SS. An overview of CCN4 (WISP1) role in human diseases. Journal of Translational Medicine. 2024;22(1):601. [PMID: 38937782] [PMCID: PMC11212430] [DOI]
25. Kharghani A, Rezaeian N, Yaghoubi A. Effect of Aerobic Training on Serum Levels of WISP1 and TNF-α and Insulin Resistance in Obese Men with Type 2 Diabetes. Journal of Applied Exercise Physiology. 2022;18(35):153-64.
26. Bahreini A, Fathi R. Comparing the Effect of Eight Weeks of Interval and Continuous Aerobic Training on Serum Levels of WISP1 and TNF-αin Overweight/0bese Girls. Journal of Sports and Biomotor Sciences. 2017;9(18):1-12.
27. Kermani S, Abbassi Daloii A, Abdi A, Saeidi A. The effect of resistance training on hormones secreted from the adipose tissue (WISP-1 and WISP-2) in obese men. Ebnesina. 2022;24(3):15-24.